Summary
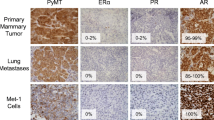
A mammary tumor cell line, designated MTCL, was successfully established from a mouse primary mammary tumor (MTP). The MTCL cells retain cytokeratin and both estrogen receptor (ER) and progesterone receptor (PR) in vitro. In vitro exposure of MTCL cells to progesterone causes a decrease in the cellular 3H-thymidine uptake, indicating an inhibition by progesterone on MTCL cellular deoxyribonucleic acid synthesis, whereas exposure of the cells to a high dose of estrogen (15 pg/ml) for 48 h causes an increase of 3H-thymidine uptake. We inoculated both MTP or MTCL tumor cells into normal cycling female, C3HeB/FeJ mice and demonstrated that the post-resection metastatic recurrence of MTCL tumors, like the original MTP tumors, depends on the time of tumor resection within the mouse estrous-cycle stage. Both MTCL and MTP tumors have similar histological appearances with the exception of less extensive tumor necrosis and higher vascularity in MTCL tumors. Equivalent levels of sex hormone receptors (ER α, ER β, and PR), epithelial growth hormone receptors (Her2/neu, EGFR1), tumor suppressors (BRCA1, P53), and cell apoptosis—relevant protein (bcl-xl) were found in these in vivo tumors by immunohistochemistry. Cyclin E protein, however, was significantly higher in MTP tumors compared with MTCL tumors. Our results indicate that MTCL cells retain many of the biologic features of the original MTP primary tumor cells, and to our knowledge, it is the first in vitro cell line that has been shown to maintain the estrous-cycle dependence of in vivo cancer metastasis.
Similar content being viewed by others

References
Allen, E. The eostrous cycle in the mouse. Am. J. Anat. 30:297–348; 1922.
Badwe, R. A.; bettelheim, R.; Millis, R. R.; Gregory, W.; Richards, M. A.; Fentiman, I. S. Cyclical tumour variations in premenopausal women with early breast cancer. Eur. J. Cancer 31A:2181–2184; 1995.
Balsari, A.; Casalini, P.; Tagliabue, E. et al. Fluctuation of HER2 expression in breast carcinomas during the menstrual cycle. Am. J. Pathol. 155; 1543–1547; 1999.
Beatson, G. On the treatment of inoperable cases of carcinoma of the mamma: suggestions for a new method of treatment with illustrative cases. Lancet 2:104–107; 1896.
Bergman, M.; Schachter, B. S.; Karelus, K.; Combatsiaris, E.; Garcia, T.; Nelson, J. Up-regulation of the uterine estrogen receptor and its messenger ribonucleic acid during the mouse estrous cycle: the role of estradiol. Endocrinology 130:1923–1930; 1992.
Bevilacqua, P.; Barbareschi, M.; Verderio, P., et al. Prognostic value of intratumoral microvessel density, a measure of tumor angiogenesis, in node-negative breast carcinoma—results of a multiparametric study. Breast Cancer Res. Treat. 36:205–217; 1995.
Bove, K.; Lincoln, D. W.; Wood, P. A.; Hrushesky, W. J.: Fertility cycle influence on surgical breast cancer cure. Breast Cancer Res. Treat. 75:65–72; 2002.
Duffy, M.; Maguire, T. M.; Hill, A.; McDermott, E.; O’Higgins, N. Metallo-proteinases: role in breast carcinogenesis, invasion and metastasis, Breast Cancer Res. 2:252–257; 2000.
Fata, J.; Chaudhary, V.; Khokha, R.; Cellular turnover in the mammary gland is correlated with systemic levels of progesterone and not 17betaestradiol during the estrous cycle. Biol. Reprod. 65:680–688; 2001.
Fisher, B.; Fisher, F.: Experimental evidence in support of the dormant tumor cell. Science 130:918–919; 1959.
Fisher, B.; Ravdin, R. G.; Ausman, R. K.; Slack, N. H.; Moore, G. E.; Noer, R. J., Surgical adjuvant chemotherapy in cancer of the breast: results of a decade of cooperative investigation. Ann. Surg. 168:337–356; 1968.
Formby, B.; Wiley, T. S., Progesterone inhibits growth and induces apoptosis in breast cancer cells: inverse effects on Bcl-2 and p53. Ann. Clin. Lab. Sci. 28:360–369; 1998.
Formby, B.; Wiley T. S., Bcl-2, survivin and variant CD44 v7–v10 are down-regulated and p53 is upregulated in breast cancer cells by progesterone: inhibition of cell growth and induction of apoptosis. Mol. Cell. Biochem. 202:53–61; 1999.
Gompel, A.; Somai, S.; Chaouat, M., et al. Hormonal regulation of apoptosis in breast cells and tissues. Steroids 65:593–598; 2000.
Hagen, A. A.; Hrushesky, W. J. Menstrual timing of breast cancer surgery. Am. J. Surg. 175:245–261; 1998.
Hambly, R.; Double, J. A.; Thompson, M. J.; Bibby, M. C. Establishment and characterisation of new cell lines from human breast tumours initially established as tumour xenografts in NMRI nude mice. Breast Cancer Res. Treat. 43:247–258; 1997.
Hirtenlehne, K.; Pec, M.; Kubista, E.; Singer, C. F. Extracellular matrix proteins influence phenotype and cytokine expression in human breast cancer cell lines. Eur. Cytokine Netw. 13:234–240;2002.
Hrushesky, W. J.; Bluming, A. Z.; Gruber S. A.; Sothern, R. B. Menstrual influence on surgical cure of breast cancer. Lancet 2:949–952; 1989.
Hrushesky, W. J.; Lester, B.; Lannin, D. Circadian coordination of cancer growth and metastatic spreal. Int. J. Cancer 83:365–373; 1999.
Kobayashi, M.; You, S,; Wood, P.; Hrushesky, W. Prominent circadian and estrous cycle impact upon tumor biology. “Circadian Rhythms and Sleep; View to the Future” Jacksonville, FL: Eighth Meeting Society for Research on Biological Rhythms; 23:2002.
Kroman, N.; Hojgaard, A.; Andersen, K. W., et al. Timing of surgery in relation to menstrual cycle does not predict the prognosis in primary breast cancer. Danish Breast Cancer Cooperative Group. Eur. J. Surg. Oncol. 20:430–435; 1994.
Laforga, J.; Aranda, F. I. Angiogenic index: a new method for assessing microvascularity in breast carcinoma with possible prognostic implications. Breast J. 6:103–107; 2000.
Michael, S.D. Plasma prolactin and progesterone during the estrous cycle in the mouse. Proc. Soc. Exp. Biol. Med. 153:254–257; 1976.
Moore, M.; Conover, J. L.; Franks, K. M. Progestin effects on long-term growth, death, and Bcl-xL in breast cancer cells. Biochem. Biophys. Res. Commun. 277:650–654; 2000.
Nomura, Y.; Kataoka, A.; Tsutsui, S.; Murakami, S.; Takenaka, Y. Lack of correlation between timing of surgery in relation to the menstrual cycle and prognosis of premenopausal patients with early breast cancer. Eur. J. Cancer 35:1326–1330; 1999.
Ory, K.; Lebeau, J.; Levalois, C.; Bishay, K.; Fouchet, P.; Allemand, I.; Therwath, A.; Chevillard, S. Apoptosis inhibition mediated by medroxy-progesterone acetate treatment of breast cancer cell lines. Breast Cancer Res. Treat. 68:187–198; 2001.
Pollock, R.; Lotzova, E. Surgical-stress-related suppression of natural killer cell activity: a possible role in tumor metastasis. Nat. Immun. Cell Growth Regul. 6:269–278; 1987.
Pollock, R.; Lotzova, E.; Stanford, S. D.; Romsdahl, M. M. Effect of surgical stress on murine natural killer cell cytotoxicity. J. Immunol. 138:171–178; 1987.
Ratajczak, H. V.; Sothern, R. B.; Hrushesky, W. J. M. Estrous influence on surgical cure of a mouse breast cancer. J. Exp. Med. 168:73–83; 1988.
Saad, Z.; Bramwell, V.; Wilson, S.; O’Malley, F.; Jeacock, J.; Chambers, A. Expression of genes that contribute to proliferative and metastatic ability in breast cancer resected during various menstrual phases. Lancet 351:1170–1173; 1998.
Vantyghem, S.; Postenka, C. O.; Chambers, A. F. Estrous cycle influences organ-specific metastasis of B16F10 melanoma cells. Cancer Res. 63:4763–4765; 2003.
Wells, W.; Rainer, R. O.; Memoli, V. A. Basic principles of image processing. Am. J. Clin. Pathol. 98:493–501; 1992.
You, S.; Yao, K.; Cao, Y. Latency of Epstein-Barr virus and its relationship to nasopharyngeal carcinomas. Zhonghua Zhongliu Zazhi 18:23–26; 1996.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
You, S., Li, W., Kobayashi, M. et al. Creation of a stable mammary tumor cell line that maintains fertility-cycle tumor biology of the parent tumor. In Vitro Cell.Dev.Biol.-Animal 40, 187–195 (2004). https://doi.org/10.1290/1543-706X(2004)40<187:COASMT>2.0.CO;2
Accepted:
Issue Date:
DOI: https://doi.org/10.1290/1543-706X(2004)40<187:COASMT>2.0.CO;2